Minnowbrook Microelectronics Conference
September 14, 2016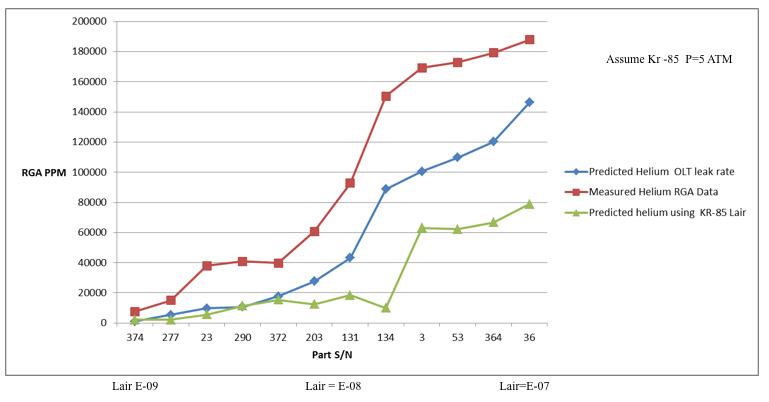
Hermeticity Testing And Issues With Correlation
September 18, 2016 IMAPS PDC: Packaging and Testing of Implanted Medical Products
IMAPS PDC: Packaging and Testing of Implanted Medical Products
I recently taught a course at the Packaging and Testing of Implanted Medical Products.
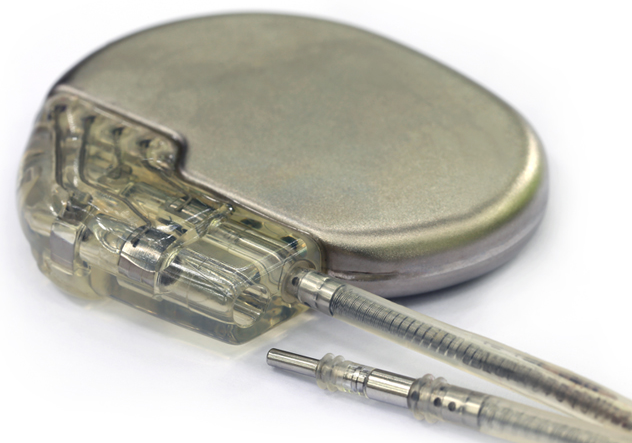
Most of the packaging technology used to manufacture implanted Class III medical products such as pacemakers, cochlear implants and IPGs, was borrowed from the Military electronics industry 40 years ago. In general, Class III medical devices are chip and wire hybrids packaged into titanium hermetic cans. A lot of the in-process quality controls and screen testing is predicated upon Mil-Std-883 Test Methods.
But the medical device industry, along with the aerospace industry, is moving away from costly, bulky hermetic packages and trying to package the devices in a “non-hermetic” sort of way. However, this requires new materials and coatings and new ways of testing the package to assure a reliable product for the expected time in vivo.
My goal with this course was to educate the attendee on the critical aspects of conventional hermetic testing and packaging and to explore and discuss the new materials and testing methods that would be practical for next generation medical implants…. hopefully you can attend and come early to participate in the IMAPS symposium and listen to technical presentations and learn more about the supplier base.
Hermetic and non-hermetic packaging and testing of microelectronics, sensors, MEMS, hybrids and microwave components for use as implanted Class II/III devices in vivo is of critical importance. Cost, reliability, small form factors, biocompatibility and patient safety are driving concerns. This course begins with an overview of traditional hermetic packaging and testing approaches that have been in use for over forty years. Most pacemakers, IPGs, cochlear implants in use today follow a prescribed and proven path of hermetic sealing and testing to assure product reliability and patient safety, which includes hermeticity testing in accordance with MIL-STD-883 Test Method 1014.
Today however, the research is directed at development of a non-hermetic package that is at least as good as the proven path. Packages made from polymeric materials require a different approach from a manufacturing and testing standpoint. The problem is now one of moisture diffusion through the barrier and package interfaces. Candidate materials such as parylene, PDMS, various ALD and CVD organic and inorganic coatings, LCP, silicones ect. are reviewed and application processes discussed. How to test and evaluate “non-hermetic” packaging methods is a primary learning objective along with an understanding of the risks and consequences of failure.