
Hermeticity: Frequently Asked Questions
September 29, 2016
Hermeticity Evaluation Testing
October 2, 2016Why Three Monolayers Of Moisture Are Important
Webinar: Hermeticity Testing For Military and Medical Microcircuits In-Plant TrainingIntroduction
The deleterious effects of moisture on microelectronic components are well-known. Failure modes involving moisture include metallization corrosion, electrical leakage/device instability, stiction in MEMs, metal migration, fogging in electro-optics, performance issues in RF microwave modules, and vacuum deterioration in packages sealed at reduced pressure.
Minimum thresholds of moisture required to activate the first four failure modes above depends on device type and design, composition and morphology of material surfaces, levels of surface cleanliness, and other factors.
It is frequently mentioned that three monolayers of adsorbed water molecules are the threshold condition for supporting electrical conduction across a surface.1-3 This kind of surface electrical effect can lead to parametric instability of devices, or galvanic action that corrodes and/or causes metal migration that physically damages critical metallization runs, contacts, or wire-bonded connections.
Three monolayers of water molecules do not seem like very much. Based on a typical diameter of about 2.8 Angstroms for a water molecule, three monolayers would be a layer of molecules only about 8 Angstroms thick. So there is a question as to how conductive three monolayers of water can be.
Electrical conduction by monolayers of adsorbed water
The composition of monolayers of water adsorbed from headspace humidity in cavity packages, in principle, should be the same as “ultrapure” (UPW) water prepared for semiconductor manufacturing by deionization. Such UPW has an electrical resistivity value of 18.25 megohm-cm at 25°C, equivalent to an electrical conductivity of 0.055 microSiemens-cm at 25°C.
This is water of theoretical purity. However, UPW has an enormous affinity for soluble ionic species. Even the tiniest quantity of an aqueous-soluble impurity will dissolve in adsorbate or condensate derived from headspace moisture, immediately raising its electrical conductivity above the value for theoretically pure water of 0.055 μS-cm. The fewer monolayers adsorbed, and assuming the surface has any available ionic species, the more conductive will be the adsorbate, since the effective concentration of ions per unit volume will be higher. Impurity ions that typically contribute to this are the alkali ions, Na+, K+, (and to a lesser extent Li+), and the halide ions, Cl–, F–, (and to a lesser extent Br–).
Exactly how conductive can such adsorbate be? Scanning Tunneling Microscopy (STM) provides the answer4. In STM, a sharp conducting probe tip is brought into very close proximity of a surface so that quantum tunneling creates an atomic-scale image of the surface. The surface must be electrically conductive for the tip to generate an image. Insulative materials cannot be directly imaged by STM. However, in a study of mica, a non-conductor, it was found that if at least one monolayer of water molecules was adsorbed over its surface, STM provided good images. Figure 1 plots conductivity current
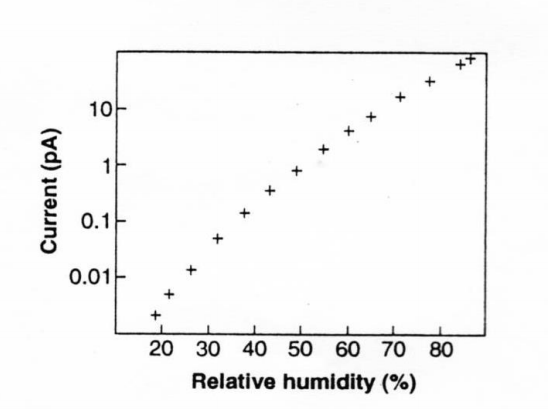
Figure 1: Electrical conductivity across mica as function of relative humidity, as determined by STM4.
across insulative mica, determined by STM, as a function of the relative humidity in which measurements were conducted. The study reports that just one monolayer of water molecules provides sufficient conductivity for STM images to be obtained because of the electrical conductivity it imparted to the surface. Figure 1 shows that the electrical conductivity of adsorbate ranges over five orders of magnitude between 20-90% RH.
STM clearly shows that only a few monolayers of adsorbate have adequate electrical conductance to initiate, sustain, and/or promote several of the moisture-oriented failure mechanisms mentioned earlier.
Headspace moisture concentration to produce three monolayers
How much headspace water vapor is needed to produce three monolayers of adsorbate? Calculations for cavity packages in the 0.01-0.05cc volume range show that about 5000ppmV (the moisture content criterion in Mil-Standards5) of headspace moisture is the threshold condition from which three monolayers of water will adsorb to interior cavity surfaces 1-3.
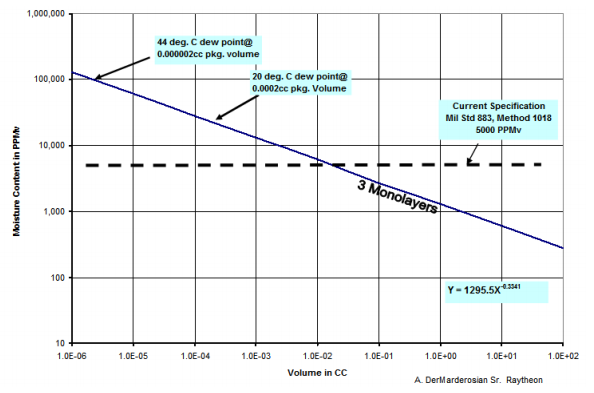
Figure 2. Three monolayer adsorption threshold as a function of moisture concentration and internal cavity volume6.
Because of surface area to volume ratio effects, the three monolayer concentration threshold is not constant over the range of volumes and declines as package volume increases.
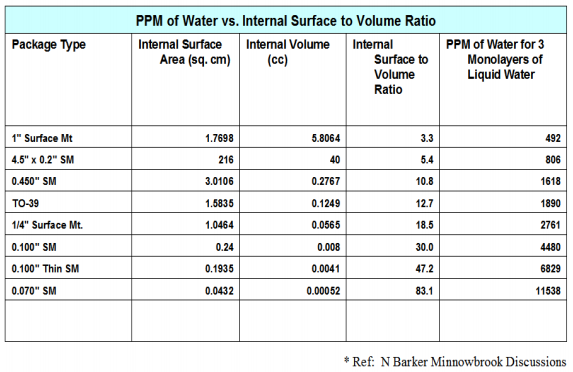
Figure 3. PPM of moisture vs. internal surface area to volume ratios for commonly used packages7.
Figure 3 shows the three monolayer calculation for a number of commonly used package styles and associated volumes. In reality moisture condensation on surfaces depends on local temperature, contamination, defects, surface energy and other types of nucleation sites which tend to cause the moisture to condense out in a non-uniform manner. In addition, some materials such as ceramics and previously dried out epoxies tend to getter the excess moisture until saturation during the onset of a condensation event.
Can 5000 ppmV headspace moisture content actually yield three adsorbed monolayers?
Two separate laboratory studies investigated adsorbed monolayers. X-ray Photoelectron Spectroscopy (XPS) was used to determine how much water adsorbed onto surfaces of gold-electroplated package lids8. Lids were exposed to varying levels of humidified gas while the O (1s) peak for oxygen in surface H2O molecules was monitored. Since XPS is conducted in vacuum, water on surfaces in this experiment was determined to be chemisorbed.
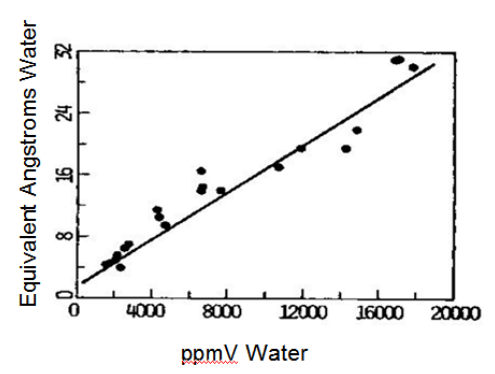
Figure 4. Thickness of water adsorbate on electroplated gold as function of headspace moisture content8.
From Fig. 4, 5000 ppmV moisture produced a water thickness on gold of about 8-12 Angstroms, corresponding to about three to four water monolayers.
In a separate study, mass changes of gold-metallized quartz crystal oscillators (QCO), conducted at 1 atm with gases of varying moisture content, was used to determine mass/unit area (hence thickness) of water physisorbed to the gold surfaces9. See Fig. 5.
Paraphrasing the authors’ discussion of Fig. 5, the onset of multilayer formation occurs at about 6000 ppmV. It would be expected this would begin at about one monolayer of water, or about 4.5A of thickness. The isotherms show that the inflection point near 5000-6000ppmV water occurs at a 4-5A increase in thickness, referenced to the zero point under dry conditions. This implies that a monolayer forms at about 5000 ppmV water.
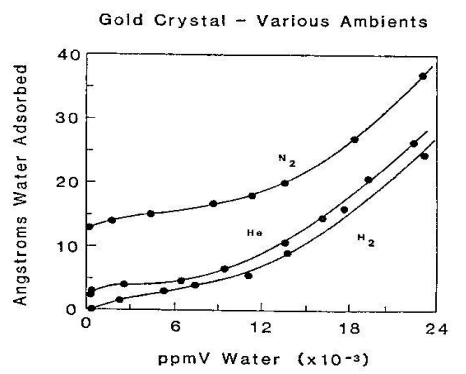
Figure 5. Thickness of water physisorbed onto gold-coated quartz crystal oscillators as function of headspace moisture content. (Note: the original manuscript contains a typographical error in the label for the ppmV Water axis: the exponent should be +3)9.
Conclusion
STM confirms that a few monolayers of water are electrically conductive. Two disparate analytical methods, XPS depth profiling and QCO weight gain measurements, confirm that about a few monolayers of water adsorb to gold surfaces from ≈5000ppmV headspace moisture content. These observations underscore the importance of controlling headspace moisture content to avoid three monolayer adsorption in any cavity package and thereby avoid moisture related failure mechanisms in the product. The fixed internal water vapor limit in Mil-Standards of 5000ppmV is roughly appropriate for cavity package volumes of 0.01-0.05cc. If all other factors are equal, the 5000ppmV limit is too stringent for smaller volumes and too lenient for larger volume enclosures.
References
1. A. DerMarderosian, Sr., “Hermeticity and Moisture Ingress”, Proceedings, RADC/NBS Workshop on Moisture Measurement and Control for Semiconductor Devices, NBSIR 84-2852, May, 1984, pp. 196-216.
2. G. Davy, :How to Raise the Permissible Leak Rate by Four Orders of Magnitude”, Proceedings, RADC/NBS Workshop on Moisture Measurement and Control for Semiconductor Devices, NBSIR 84-2852, May, 1984, pp. 180-195.
3. R.P. Merrett, “A Simple Method of Using the Die of an Integrated Circuit to Measure the Relative Humidity Inside Its Encapsulation”, Proceedings, International Reliability Physics Symposium, 1980, pp. 17-25.
4. R. Guckenberger, et. al., “Scanning Tunneling Microscopy of Insulators and Biological Specimens Based on Lateral Conductivity of Ultrathin Water Films”, Science, vol. 266, 2 December, 1994, pp. 1538-1540, and additional references therein.
5. Test Method 1018.6, Mil-Standard 883H, and other military standard documents.
6. A. DerMarderosian, Sr., private communication, Minnowbrook Microelectronics Conference, 2007.
7. N. Barker, private communication, Minnowbrook Microelectronics Conference, 2005.
8. W.E. Swartz, et. al., “The Adsorption of Water on Metallic Packages”, Proceedings, International Reliability Physics Symposium, 1983, pp. 52-56.
9. J.M. Ammons, et. al., “The Physisorption of Water Onto Integrated Circuit Package Components”, Proceedings, RADC/NBS Workshop on Moisture Measurement and Control for Semiconductor Devices, NBSIR 84-2852, May, 1984, pp. 226-235.
About The Authors
Tom Green, T.J. Green Associates, LLC
739 Redfern Lane, Bethlehem, PA 18017, USA
Phone: 610-625-2158
Email: [email protected]
www.tjgreenllc.com
Bob Lowry, Electronic Materials Consultant
3000 Purple Martin Lane, Indialantic, FL 32903, USA
Phone: 321-777-9949
Email: [email protected]
www.electronic-materials.com



